Chemistry, 22.03.2021 22:30 Solomon4148
Green plants use light from the Sun to drive photosynthesis, a chemical reaction in which liquid water and carbon dioxide gas form aqueous glucose C6H12O6 and oxygen O2 gas. Calculate the moles of water needed to produce 0.075mol of glucose. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3


Chemistry, 23.06.2019 14:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 14.0 mol cesium fluoride with 14.0 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1
You know the right answer?
Green plants use light from the Sun to drive photosynthesis, a chemical reaction in which liquid wat...
Questions

English, 30.05.2020 14:58

Chemistry, 30.05.2020 14:58




Biology, 30.05.2020 14:58



Mathematics, 30.05.2020 14:58

Mathematics, 30.05.2020 14:58


Computers and Technology, 30.05.2020 14:58








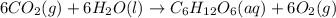
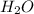
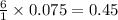 moles of
moles of