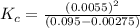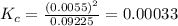Chemistry, 22.03.2021 20:10 xcrysttallx
QUESTION 6 Consider the following reaction between the diatomic and monatomic forms of iodine: I2 (g) <-> 2I (g) When 0.095 M I2 is initially placed in a previously empty container and sealed, the system slowly reaches equilibrium. When equilibrium is reached, it is found that there is an equilibrium concentration of 0.0055 M of the monatomic form of iodine. Calculate the (unitless) equilibrium constant Kc. Round your answer to two sig figs, and express it in scientific notation.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1

You know the right answer?
QUESTION 6 Consider the following reaction between the diatomic and monatomic forms of iodine: I2 (g...
Questions


Social Studies, 09.07.2019 19:00





Chemistry, 09.07.2019 19:00



Mathematics, 09.07.2019 19:00


Mathematics, 09.07.2019 19:00

Health, 09.07.2019 19:00


Arts, 09.07.2019 19:00



Social Studies, 09.07.2019 19:00

Mathematics, 09.07.2019 19:00

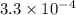
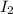 = 0.095 M
= 0.095 M 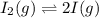
![K_c=\frac{[l]^2}{[I_2]}](/tpl/images/1211/7332/312cd.png)