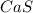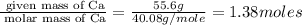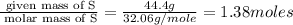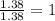Chemistry, 21.03.2021 07:50 itsgiovanna
What is the formula of the compound between calcium and sulfur that has the percent composition 55.6% (by mass) calcium and 44.4% (by mass) sulfur? What is the formula of the compound between calcium and sulfur that has the percent composition 55.6% (by mass) calcium and 44.4% (by mass) sulfur? Ca2S4 CaS Ca2S CaS4 CaS2 Request

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
You know the right answer?
What is the formula of the compound between calcium and sulfur that has the percent composition 55.6...
Questions

History, 26.08.2019 13:30


Chemistry, 26.08.2019 13:30



Physics, 26.08.2019 13:30


Social Studies, 26.08.2019 13:30

Social Studies, 26.08.2019 13:30



History, 26.08.2019 13:30


History, 26.08.2019 13:30

History, 26.08.2019 13:30


English, 26.08.2019 13:30



Mathematics, 26.08.2019 13:30