
Chemistry, 20.03.2021 03:00 liyahheadhigh
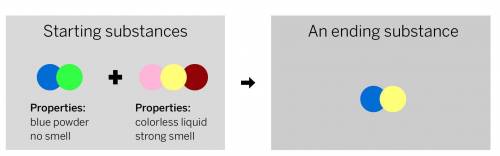
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a strong smell. Their repeating groups of atoms are shown above on the left. After they were mixed, the chemist analyzed the results and found two substances. One ending substance had the repeating group of atoms shown above on the right. Is the ending substance the same substance as the blue powder? What happened to the atoms of the starting substances when the ending substances formed? Be sure to explain your answers to both of these questions.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

You know the right answer?
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a s...
Questions


Mathematics, 22.05.2020 02:06




Mathematics, 22.05.2020 02:06





History, 22.05.2020 02:06

English, 22.05.2020 02:06

Mathematics, 22.05.2020 02:06


Chemistry, 22.05.2020 02:06





Mathematics, 22.05.2020 02:06



