
Chemistry, 19.03.2021 22:00 destinywashere101
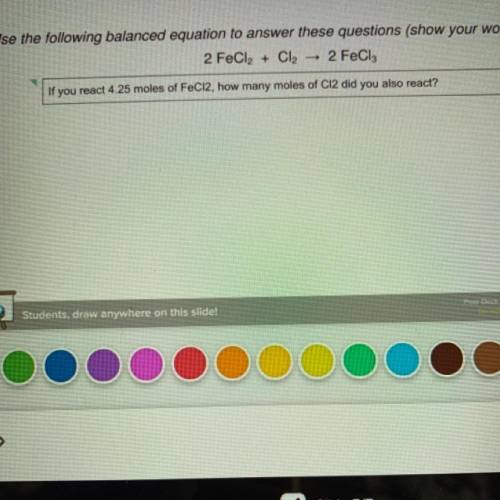
PLEASE HELP I WILL MARK YOU BRAINLIEST !!If you react 4.25 moles of FeCl2, how many moles of Cl2 did you also react? ILL GIVE 12 POINTS TOO PLEASE IM BEGGING FOR HELP

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
PLEASE HELP I WILL MARK YOU BRAINLIEST !!If you react 4.25 moles of FeCl2, how many moles of Cl2 did...
Questions






Chemistry, 03.09.2019 04:10




Medicine, 03.09.2019 04:10












