Which compound is more soluble in water at 25°C?
A.
MgF2 (Ksp = 5.2 x 10-11)
B.
S...

Chemistry, 19.03.2021 22:00 janiadavis35
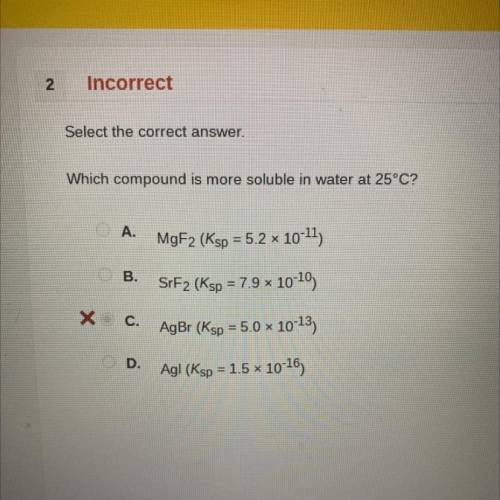
Which compound is more soluble in water at 25°C?
A.
MgF2 (Ksp = 5.2 x 10-11)
B.
SrF2 (Ksp = 7.9 x 10-10)
C.
AgBr (Ksp = 5.0 x 10-13)
D.
Agl (Ksp = 1.5 * 10-16)

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

You know the right answer?
Questions














Arts, 29.10.2019 21:31

History, 29.10.2019 21:31

Social Studies, 29.10.2019 21:31

History, 29.10.2019 21:31





