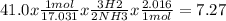Please answer soon
Consider the following balanced chemical reaction:
N2 + 3 H2 → 2 NH3...

Chemistry, 19.03.2021 20:10 tdahna0403
Please answer soon
Consider the following balanced chemical reaction:
N2 + 3 H2 → 2 NH3
If you needed to produce 41.0 g of NH3, how many grams of H2 would you need?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3


Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
Questions



SAT, 11.01.2021 18:20

Mathematics, 11.01.2021 18:20


Mathematics, 11.01.2021 18:20

Mathematics, 11.01.2021 18:20

Mathematics, 11.01.2021 18:20

Business, 11.01.2021 18:20

Engineering, 11.01.2021 18:20

Mathematics, 11.01.2021 18:20

Physics, 11.01.2021 18:20



Biology, 11.01.2021 18:20



Business, 11.01.2021 18:20

Mathematics, 11.01.2021 18:20