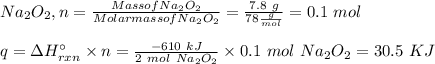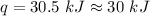Two trials are run, using excess water. In the first trial, 7.8 g of Na2O2(s) (molar mass 78 g/mol) is mixed with 3.2 g of S(s). In the second trial, 7.8 g of Na2O2(s) is mixed with 6.4 g of S(s). The Na2O2(s) and S(s) react as completely as possible. Both trials yield the same amount of SO2(aq). Which of the following identifies the limiting reactant and the heat released, q, for the two trials at 298 K?
Limiting Reactant q
A. S 30. kJ
B. S 61 kJ
C. Na2O2 30. kJ
D. Na2S2 61 kJ

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2
You know the right answer?
Two trials are run, using excess water. In the first trial, 7.8 g of Na2O2(s) (molar mass 78 g/mol)...
Questions







Mathematics, 13.11.2020 22:10

Mathematics, 13.11.2020 22:10

English, 13.11.2020 22:10






Advanced Placement (AP), 13.11.2020 22:10

English, 13.11.2020 22:10

Mathematics, 13.11.2020 22:10


Mathematics, 13.11.2020 22:10

History, 13.11.2020 22:10

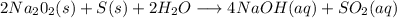
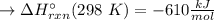
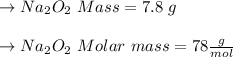
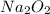 Has been the reactant which is limited since the two experiments are equal to
Has been the reactant which is limited since the two experiments are equal to