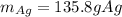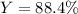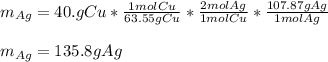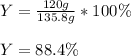Chemistry, 19.03.2021 14:00 squawk1738
Cu + 2AgNO3 → Cu(NO3)2 + 2Ag If a 40. g sample of Copper is used with an excess of silver nitrate, calculate the theoretical yield of silver. What is the percent yield of the silver, if 120 g was collected?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 23.06.2019 01:10
A5.00 g of a in . g of at aa 5.00 g of b in . g of .?at .
Answers: 1

Chemistry, 23.06.2019 02:50
Select the correct location on the image identify the element that humans need to breathe. 2015 er r ights reserved
Answers: 3
You know the right answer?
Cu + 2AgNO3 → Cu(NO3)2 + 2Ag
If a 40. g sample of Copper is used with an excess of silver nitrate,...
Questions


Mathematics, 12.02.2021 06:30


Health, 12.02.2021 06:30

Mathematics, 12.02.2021 06:30


Mathematics, 12.02.2021 06:30


Mathematics, 12.02.2021 06:30

Mathematics, 12.02.2021 06:30

Mathematics, 12.02.2021 06:30

Health, 12.02.2021 06:30


Mathematics, 12.02.2021 06:30



English, 12.02.2021 06:30



Mathematics, 12.02.2021 06:30