Chemistry, 19.03.2021 05:50 taufajane3887
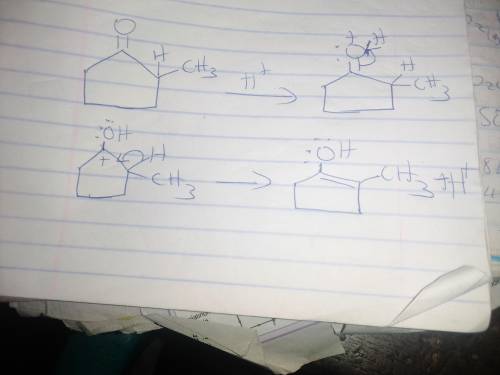
When optically active (S)-2-methylcyclopentanone is treated with an acid (H3O ), the compound loses its optical activity. Explain this observation and draw a mechanism that shows how racemization occurs. For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products. Do not use abbreviations such as Me or Ph.

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3
You know the right answer?
When optically active (S)-2-methylcyclopentanone is treated with an acid (H3O ), the compound loses...
Questions



Mathematics, 23.09.2019 18:30

Mathematics, 23.09.2019 18:30

Mathematics, 23.09.2019 18:30


Mathematics, 23.09.2019 18:30




Mathematics, 23.09.2019 18:30

English, 23.09.2019 18:30



Arts, 23.09.2019 18:30

Mathematics, 23.09.2019 18:30

History, 23.09.2019 18:30


History, 23.09.2019 18:30