
Chemistry, 18.03.2021 20:30 titowanthony16
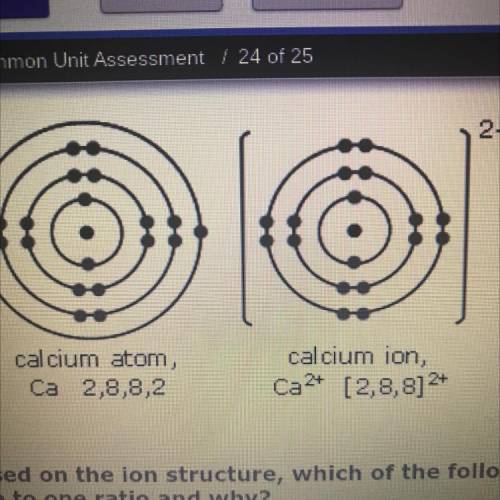
Based on the ion structure, which of the following elements would form a bond with calcium in a
one to one ratio and why?
O A. One bromine ion would bond to one calcium ion because the combination of their charges would result
in a full octet.
OB. One bromine ion would bond to one calcium ion because they are both located on period 4.
C. One selenium ion would bond to one calcium ion because they are both located on period 4.
D. One selenium ion would bond to one calcium ion because the combination of their charges would result
in a full octet.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1

Chemistry, 23.06.2019 00:30
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2
You know the right answer?
Based on the ion structure, which of the following elements would form a bond with calcium in a
one...
Questions


Mathematics, 11.11.2020 19:00


English, 11.11.2020 19:00


English, 11.11.2020 19:00


English, 11.11.2020 19:00

Mathematics, 11.11.2020 19:00


Business, 11.11.2020 19:00



Physics, 11.11.2020 19:00

Mathematics, 11.11.2020 19:00


English, 11.11.2020 19:00


Mathematics, 11.11.2020 19:00



