Chemistry, 18.03.2021 01:50 taylor3932
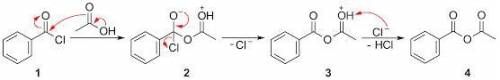
Draw a mechanism for this reaction. In the reaction scheme, an acyl chloride reacts with an acid to give an acid anhydride. The acyl chloride contains a central C atom that has a double bond to O, a single bond to Cl, and a single bond to a benzene ring. The acid is CH3COH with an O atom double-bonded to the second (from left to right) carbon. The produced anhydride is COCCH3 with a benzene ring attached to the first (from left to right) carbon and an O atom double-bonded to the firth and the second carbons.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 01:40
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1

Chemistry, 22.06.2019 21:20
The organs inside the body and how they function together
Answers: 3

Chemistry, 23.06.2019 06:50
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1
You know the right answer?
Draw a mechanism for this reaction. In the reaction scheme, an acyl chloride reacts with an acid to...
Questions


History, 27.09.2019 11:00


Chemistry, 27.09.2019 11:00

Social Studies, 27.09.2019 11:00


History, 27.09.2019 11:00




Mathematics, 27.09.2019 11:00



Business, 27.09.2019 11:00

English, 27.09.2019 11:00

History, 27.09.2019 11:00

History, 27.09.2019 11:00

Biology, 27.09.2019 11:00


History, 27.09.2019 11:00