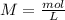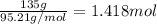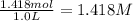Chemistry, 18.03.2021 01:30 markrandsdenn
If 135 grams of MgCl2 are dissolved in a 1.0 L solutions, what is the molarity of the solution?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1


Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3
You know the right answer?
If 135 grams of MgCl2 are dissolved in a 1.0 L solutions, what is the molarity of the solution?...
Questions




Health, 27.01.2021 19:50

Health, 27.01.2021 19:50

Mathematics, 27.01.2021 19:50

English, 27.01.2021 19:50

Mathematics, 27.01.2021 19:50







Mathematics, 27.01.2021 19:50


English, 27.01.2021 19:50


History, 27.01.2021 19:50