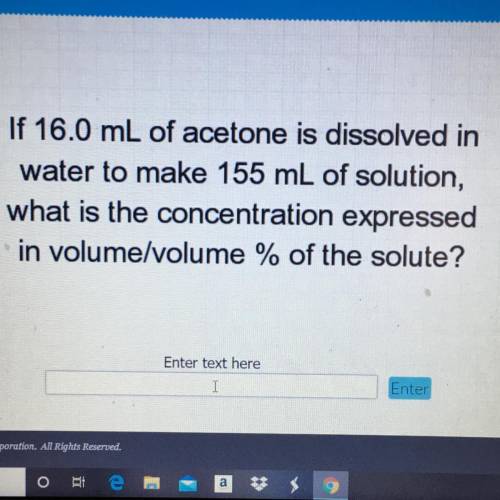
If 16.0 mL of acetone is dissolved in
water to make 155 mL of solution,
what is the concentra...

Chemistry, 18.03.2021 01:20 janely6017
If 16.0 mL of acetone is dissolved in
water to make 155 mL of solution,
what is the concentration expressed
in volume/volume % of the solute?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3
You know the right answer?
Questions



Mathematics, 11.08.2021 02:40


Mathematics, 11.08.2021 02:40

Biology, 11.08.2021 02:40

Mathematics, 11.08.2021 02:40


Biology, 11.08.2021 02:40



Mathematics, 11.08.2021 02:50

Mathematics, 11.08.2021 02:50


Mathematics, 11.08.2021 02:50

Mathematics, 11.08.2021 02:50

Mathematics, 11.08.2021 02:50

Mathematics, 11.08.2021 02:50

World Languages, 11.08.2021 02:50

Mathematics, 11.08.2021 02:50



