
Chemistry, 18.03.2021 01:20 alondra6190
Nitrogen dioxide gas is dark brown in color and remains in equilibrium with dinitrogen tetroxide gas, which is colorless.
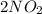 (g) ⇌
(g) ⇌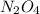 (g)
(g)
When the mixture was moved from room temperature to a higher temperature, the mixture turned darker brown. Which of the following conclusions about the mixture is true?
A. The forward reaction is exothermic.
B. The backward reaction has a negative net enthalpy.
C. The colorless gas has a higher enthalpy than the brown gas.
I know that is an exothermic reaction, but it shifts to left, so it should be a backward reaction, not a forward. Therefore, I don't think it is A. I am leaning towards B, but I would like someone to double-check my thinking. Please no fake answers or external links. Thanks in advance!

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3
You know the right answer?
Nitrogen dioxide gas is dark brown in color and remains in equilibrium with dinitrogen tetroxide gas...
Questions












Mathematics, 01.10.2019 16:20





Social Studies, 01.10.2019 16:20





