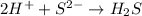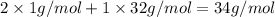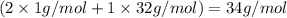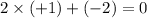During all chemical reactions, mass, energy, and charge are
(1) absorbed (3) formed
(2)...

Chemistry, 03.11.2019 18:31 angelteddy033
During all chemical reactions, mass, energy, and charge are
(1) absorbed (3) formed
(2) conserved (4) released

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 01:40
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2

Chemistry, 22.06.2019 04:30
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1
You know the right answer?
Questions

Advanced Placement (AP), 15.12.2020 18:10


Mathematics, 15.12.2020 18:10




Mathematics, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10

English, 15.12.2020 18:10

History, 15.12.2020 18:10

Social Studies, 15.12.2020 18:10


Physics, 15.12.2020 18:10

Social Studies, 15.12.2020 18:10


Mathematics, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10


Mathematics, 15.12.2020 18:10