
Chemistry, 13.03.2021 04:50 genyjoannerubiera
Text 8.7 Using the virial equation of state for hydrogen at 298 K given in problem 7 (text 8.6), calculate a. The fugacity of hydrogen at 500 atm and 298 K b. The pressure at which they fugacity is twice the pressure c. The change in Gibbs free energy caused by a compression of 1 mole of hydrogen from 1 to 500 atm. What is the magnitude of the contribution to (c) caused by the non ideality of hydrogen

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2


Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3
You know the right answer?
Text 8.7 Using the virial equation of state for hydrogen at 298 K given in problem 7 (text 8.6), cal...
Questions


Spanish, 05.11.2020 20:40

Mathematics, 05.11.2020 20:40

Mathematics, 05.11.2020 20:40


Biology, 05.11.2020 20:40

Chemistry, 05.11.2020 20:40

Geography, 05.11.2020 20:40

English, 05.11.2020 20:40


Mathematics, 05.11.2020 20:40

Mathematics, 05.11.2020 20:40



Advanced Placement (AP), 05.11.2020 20:40



Arts, 05.11.2020 20:40

Mathematics, 05.11.2020 20:40

Mathematics, 05.11.2020 20:40

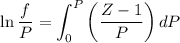
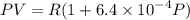 ........(1)
........(1)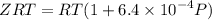
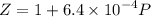
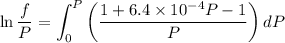
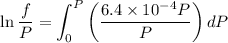
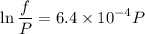
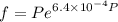
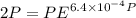
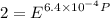
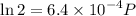
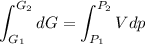
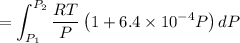
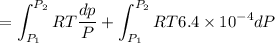
![$\Delta G=R[\ln\frac{P_2}{P_1}+6.4 \times 10^{-4}(P_2-P_1)]$](/tpl/images/1193/5881/be85b.png)
![$\Delta G=8.314\times 298[\ln\frac{500}{1}+6.4 \times 10^{-4}(500-1)]$](/tpl/images/1193/5881/16a41.png)


