
Chemistry, 13.03.2021 04:00 youngcie04
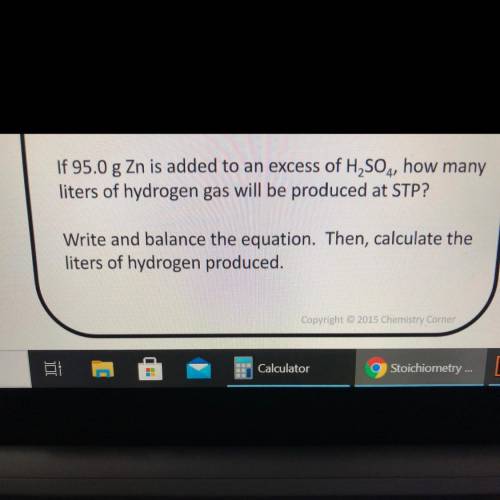
If 95.0 g Zn is added to an excess of H2SO4, how many
liters of hydrogen gas will be produced at STP?
Write and balance the equation. Then, calculate the
liters of hydrogen produced.
Please help

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 08:00
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 23.06.2019 12:20
Describe the structure of ammonium lauryl sulfate. refer to the given diagram. your answer should include the type of bonding, the elements contained, and the size and shape of the molecule. write a short paragraph.
Answers: 3
You know the right answer?
If 95.0 g Zn is added to an excess of H2SO4, how many
liters of hydrogen gas will be produced at ST...
Questions

Mathematics, 10.02.2021 01:00


Mathematics, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00

Health, 10.02.2021 01:00


Mathematics, 10.02.2021 01:00

Social Studies, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00


Mathematics, 10.02.2021 01:00

Arts, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00

Mathematics, 10.02.2021 01:00


Mathematics, 10.02.2021 01:00


Chemistry, 10.02.2021 01:00



