
Chemistry, 13.03.2021 01:00 mvtthewisdead
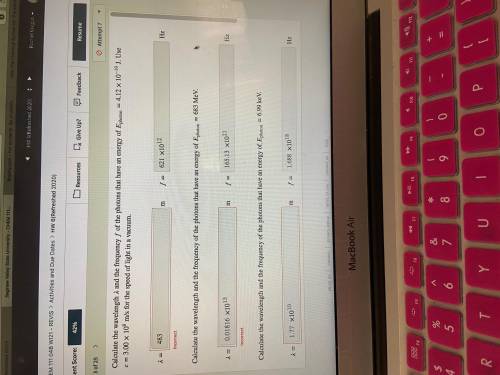
Calculate the wavelength and the frequency of the photons that have an energy of photon=4.12×10−19 J. Use =3.00×108 m/s for the speed of light in a vacuum.
Calculate the wavelength and the frequency of the photons that have an energy of photon=683 MeV.
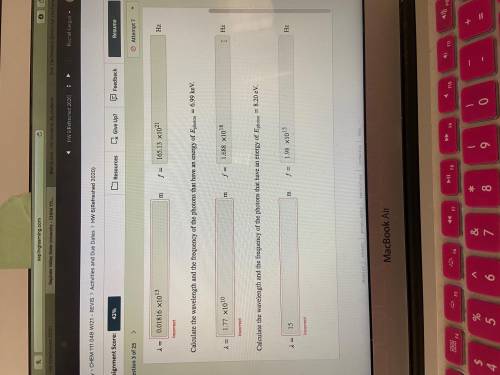
Calculate the wavelength and the frequency of the photons that have an energy of photon=6.99 keV.
Calculate the wavelength and the frequency of the photons that have an energy of photon=8.20 eV.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

You know the right answer?
Calculate the wavelength and the frequency of the photons that have an energy of photon=4.12×10−19 J...
Questions

Computers and Technology, 18.04.2020 06:19

English, 18.04.2020 06:19



Mathematics, 18.04.2020 06:19

Chemistry, 18.04.2020 06:19

Mathematics, 18.04.2020 06:19

Mathematics, 18.04.2020 06:19



History, 18.04.2020 06:19

Chemistry, 18.04.2020 06:19

English, 18.04.2020 06:19

History, 18.04.2020 06:19



Mathematics, 18.04.2020 06:20


Mathematics, 18.04.2020 06:20



