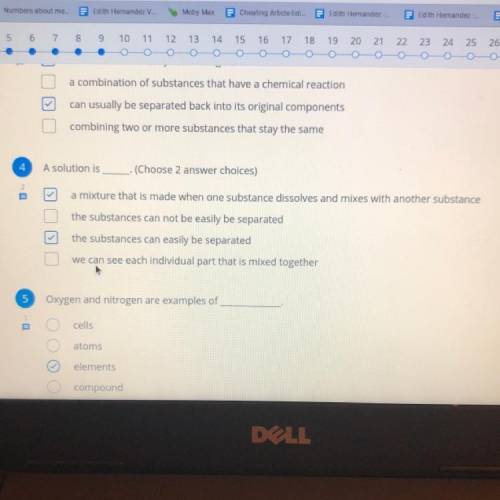
Need help with question 4.) hurry please ASAP
...

Answers: 3
Another question on Chemistry



Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
Questions

Mathematics, 23.10.2019 21:00


Mathematics, 23.10.2019 21:00



History, 23.10.2019 21:00

Mathematics, 23.10.2019 21:00




History, 23.10.2019 21:00

History, 23.10.2019 21:00

Mathematics, 23.10.2019 21:00



Mathematics, 23.10.2019 21:00


Mathematics, 23.10.2019 21:00

English, 23.10.2019 21:00