
Chemistry, 11.03.2021 20:00 07corcum85504
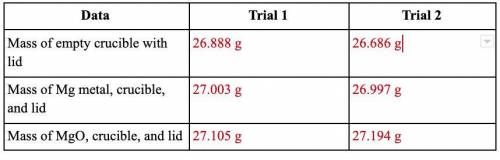
40 Points! Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for each trial.
Equation: Mg + O2 → MgO
This is the actual yield of magnesium oxide for each trial.
Trial 1: 0.217 g
Trial 2: 0.508 g

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 23.06.2019 03:00
Which of the following is a chemical property of water at 4 c
Answers: 2

You know the right answer?
40 Points! Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of...
Questions

Chemistry, 15.02.2021 08:20

History, 15.02.2021 08:20

Mathematics, 15.02.2021 08:20

English, 15.02.2021 08:20

English, 15.02.2021 08:20


Mathematics, 15.02.2021 08:20

Chemistry, 15.02.2021 08:20




English, 15.02.2021 08:20

History, 15.02.2021 08:20

Mathematics, 15.02.2021 08:20

Chemistry, 15.02.2021 08:20



History, 15.02.2021 08:20




