35 POINTS
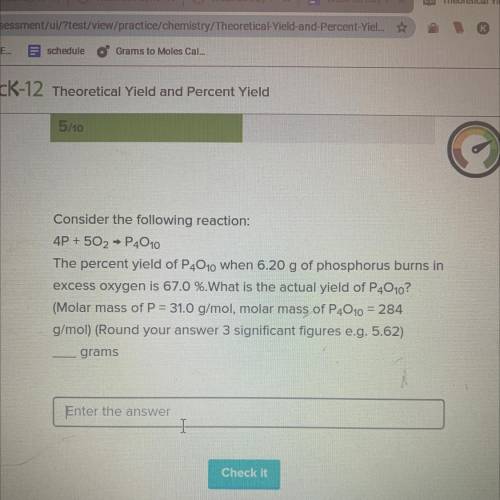
Consider the following reaction:
4P + 502 → P4010
The percent yield of P4010...

Chemistry, 11.03.2021 19:30 dontcareanyonemo
35 POINTS
Consider the following reaction:
4P + 502 → P4010
The percent yield of P4010 when 6.20 g of phosphorus burns in
excess oxygen is 67.0 %.What is the actual yield of P4010?
(Molar mass of P = 31.0 g/mol, molar mass of P4010 = 284
g/mol) (Round your answer 3 significant figures e. g. 5.62)
grams

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2


Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Questions


Social Studies, 07.10.2019 10:30


Chemistry, 07.10.2019 10:30

Mathematics, 07.10.2019 10:30

Mathematics, 07.10.2019 10:30


Mathematics, 07.10.2019 10:30





Business, 07.10.2019 10:30

English, 07.10.2019 10:30






History, 07.10.2019 10:30



