
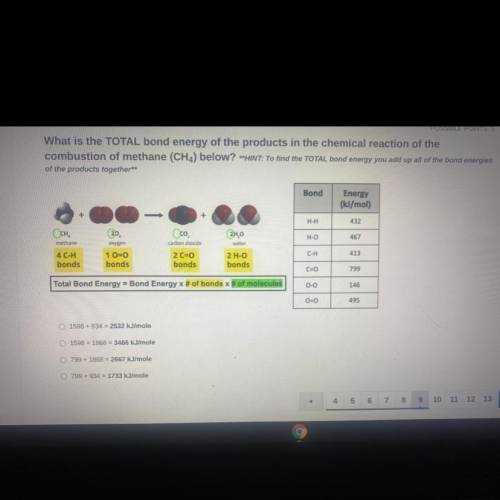
What is the TOTAL bond energy of the products in the chemical reaction of the
combustion of methane (CH4) below? **HINT: To find the TOTAL bond energy you add up all of the bond energies
of the products together**
Bond
Energy
(kJ/mol)
H-H
432
Осн.
H-O
467
methane
Oco
carbon dioxide
2 C=0
bonds
2H,0
water
2 H-O
bonds
oxygen
1 0=0
bonds
413
C-H
4 C-H
bonds
C=0
799
Total Bond Energy = Bond Energy x # of bonds of molecules
0-0
146
00
495
1598 +934 = 2532 kJ/mole
1598 +1868 = 3466 kJ/mole
799 + 1868 = 2667
kJ/mole
799 + 934 = 1733 kJ/mole

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 06:00
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
What is the TOTAL bond energy of the products in the chemical reaction of the
combustion of methane...
Questions



Mathematics, 06.05.2020 18:04





English, 06.05.2020 18:04

Mathematics, 06.05.2020 18:04

Mathematics, 06.05.2020 18:04

Mathematics, 06.05.2020 18:04



History, 06.05.2020 18:04

Mathematics, 06.05.2020 18:04








