
Chemistry, 09.03.2021 02:50 quincyjosiah07
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs out 32.mg of oxalic acid H2C2O4, a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in 250.mL of distilled water. The student then titrates the oxalic acid solution with his sodium hydroxide solution. When the titration reaches the equivalence point, the student finds he has used 60.9mL of sodium hydroxide solution. Calculate the molarity of the student's sodium hydroxide solution. Round your answer to 2 significant digits. M

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

You know the right answer?
A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs o...
Questions


Biology, 19.05.2020 15:12

Mathematics, 19.05.2020 15:12

Mathematics, 19.05.2020 15:12

English, 19.05.2020 15:12

English, 19.05.2020 15:12





Mathematics, 19.05.2020 15:12


History, 19.05.2020 15:12




History, 19.05.2020 15:13



Mathematics, 19.05.2020 15:13

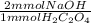 = 0.72 mmol NaOH
= 0.72 mmol NaOH

