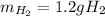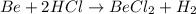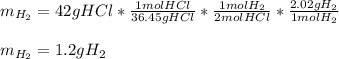Chemistry, 08.03.2021 20:10 autumn452392
If 42 grams of HCl reacts, what is the theoretical yield of hydrogen gas produced?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3


Chemistry, 22.06.2019 23:30
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3
You know the right answer?
If 42 grams of HCl reacts, what is the theoretical yield of hydrogen gas produced?...
Questions



Social Studies, 24.11.2020 22:10

Mathematics, 24.11.2020 22:10

Geography, 24.11.2020 22:10

Physics, 24.11.2020 22:10


Biology, 24.11.2020 22:10

Computers and Technology, 24.11.2020 22:10

Social Studies, 24.11.2020 22:10



Mathematics, 24.11.2020 22:10


Social Studies, 24.11.2020 22:10



Mathematics, 24.11.2020 22:10

History, 24.11.2020 22:10