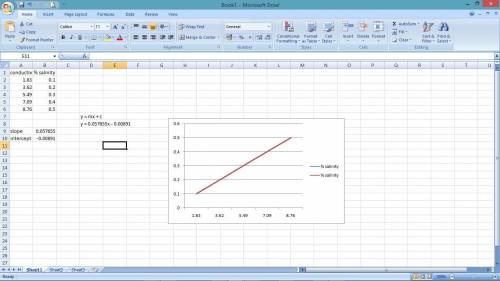
In chemistry lab student is determining the salinity of seawater using a conductivity meter. The student made standards of NaCl solutions and recorded the following data:
% Salinity Conductivity (ms)
0.10 1.83
0.20 3.62
0.30 5.49
0.40 7.09
0.50 8.76
The conductance of a 10 mL sample of unknown was found to be 5.82 ms.
Required:
a. Prepare a standard curve using the data above.
b. Determine the concentration of the unknown salt solution using the standard curve.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Apure solvent has a vapor pressure the vapor pressure of a solution. a. equal to b. lower than c. higher than
Answers: 1

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
In chemistry lab student is determining the salinity of seawater using a conductivity meter. The stu...
Questions


Mathematics, 26.03.2020 21:12

Health, 26.03.2020 21:12


Computers and Technology, 26.03.2020 21:12



English, 26.03.2020 21:12

Computers and Technology, 26.03.2020 21:12





Mathematics, 26.03.2020 21:12


Mathematics, 26.03.2020 21:12

English, 26.03.2020 21:13

Physics, 26.03.2020 21:13


Biology, 26.03.2020 21:13