
Chemistry, 06.03.2021 04:10 jetskiinbunny31
4 Al + 3 O₂ --> 2 Al2Ol₃ Given 6 moles of aluminum, how many grams of aluminum oxide will be produced?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1


Chemistry, 23.06.2019 12:10
What is the correct name for hg(no3)2? mercury (i) nitrate mercury (ii) nitrate mercury nitroxide mercury dinitride
Answers: 1
You know the right answer?
4 Al + 3 O₂ --> 2 Al2Ol₃ Given 6 moles of aluminum, how many grams of aluminum oxide will be prod...
Questions

Mathematics, 05.05.2020 08:00




History, 05.05.2020 08:00


English, 05.05.2020 08:00

English, 05.05.2020 08:00

Mathematics, 05.05.2020 08:00



Biology, 05.05.2020 08:00


History, 05.05.2020 08:00





Arts, 05.05.2020 08:01

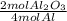 = 3 mol Al₂O₃
= 3 mol Al₂O₃

