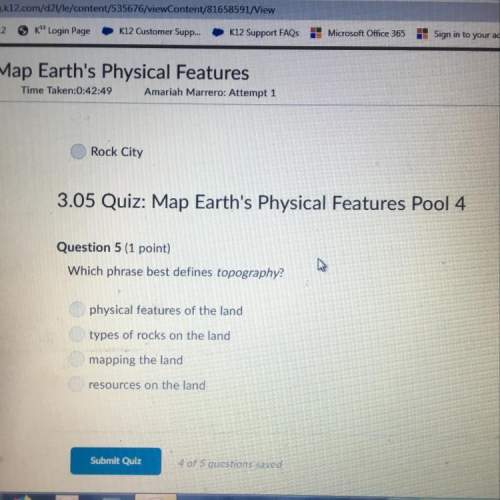
PLEASE PLEASE HELP QUICK lots of points please
Answer the following question:
Calculate...

Chemistry, 06.03.2021 01:00 jojoblue2004
PLEASE PLEASE HELP QUICK lots of points please
Answer the following question:
Calculate the mass of 45.0 L of NH3 at 87.0° C and 750. mm Hg.
Use the Ideal Gas Law formula and here are values for R:
8.134 (L * kPa)/(mol * K)
0.0821 (atm * L)/(mol * K)
62.364 (L * mmHg)/(mol * K)
Include the following with your
Which Gas Law constant did you use or which "R" value did you use? Why?
The numerical answer to the question.
An explanation of the correct number of significant figures you will use for the numerical answer.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 04:30
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1
You know the right answer?
Questions

History, 13.12.2021 17:00


Mathematics, 13.12.2021 17:00

English, 13.12.2021 17:00



Mathematics, 13.12.2021 17:00


History, 13.12.2021 17:00







English, 13.12.2021 17:00

English, 13.12.2021 17:00


Mathematics, 13.12.2021 17:00