
Chemistry, 05.03.2021 22:40 alexandra2442
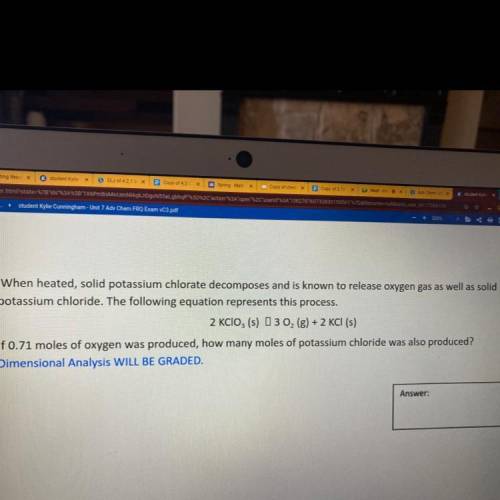
When heated, solid potassium chlorate decomposes and is known to release oxygen gas as well as solid
potassium chloride. The following equation represents this process.
2 KClO3 (s) 3 02 (g) + 2 KCl (s)
If 0.71 moles of oxygen was produced, how many moles of potassium chloride was also produced?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
You know the right answer?
When heated, solid potassium chlorate decomposes and is known to release oxygen gas as well as solid...
Questions

Mathematics, 20.09.2019 06:00

Mathematics, 20.09.2019 06:00


Chemistry, 20.09.2019 06:00





Business, 20.09.2019 06:00


Mathematics, 20.09.2019 06:00

Biology, 20.09.2019 06:00

Physics, 20.09.2019 06:00






Mathematics, 20.09.2019 06:10



