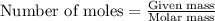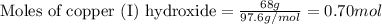Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 23:30
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3

You know the right answer?
How many moles are in 68 grams of copper (ll) hydroxide, cu(oh)2...
Questions

Health, 01.06.2021 22:20



Mathematics, 01.06.2021 22:20

Chemistry, 01.06.2021 22:20



Social Studies, 01.06.2021 22:20




Mathematics, 01.06.2021 22:30

Health, 01.06.2021 22:30


Physics, 01.06.2021 22:30

Chemistry, 01.06.2021 22:30


English, 01.06.2021 22:30


Geography, 01.06.2021 22:30