Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
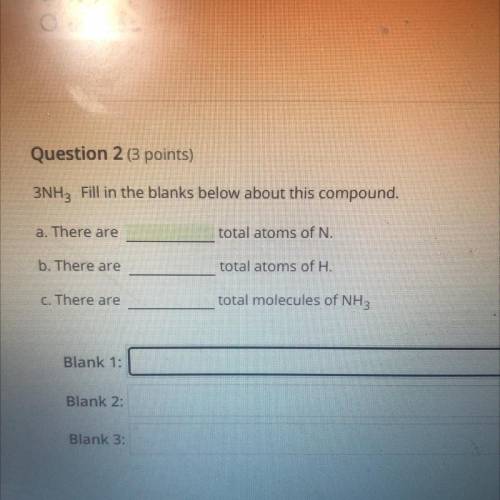
Fill in the blanks. 3NH3
...
...
Questions

Mathematics, 25.07.2019 14:10


Chemistry, 25.07.2019 14:10

Mathematics, 25.07.2019 14:10

Mathematics, 25.07.2019 14:10


Physics, 25.07.2019 14:10


English, 25.07.2019 14:10

Social Studies, 25.07.2019 14:10



Social Studies, 25.07.2019 14:10

Business, 25.07.2019 14:10


History, 25.07.2019 14:10



Mathematics, 25.07.2019 14:10