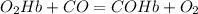Chemistry, 18.12.2019 23:31 shinekamui
According to the chemical reaction, if there are 4.5 mol of oxygenated hemoglobin present in an excess of carbon monoxide, how many moles of hemoglobin would release oxygen and bind to carbon monoxide? explain your answer.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3
You know the right answer?
According to the chemical reaction, if there are 4.5 mol of oxygenated hemoglobin present in an exce...
Questions


English, 27.05.2020 18:03





Mathematics, 27.05.2020 18:03

Mathematics, 27.05.2020 18:03

History, 27.05.2020 18:03

Mathematics, 27.05.2020 18:03

Mathematics, 27.05.2020 18:03

Mathematics, 27.05.2020 18:03

Mathematics, 27.05.2020 18:03


Mathematics, 27.05.2020 18:03


Chemistry, 27.05.2020 18:03

Chemistry, 27.05.2020 18:03


History, 27.05.2020 18:03