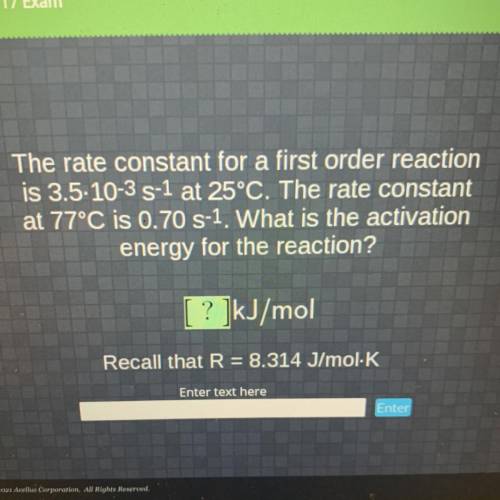
The rate constant for a first order reaction
is 3.5-10-3 S-1 at 25°C. The rate constant
at 77...

Chemistry, 03.03.2021 22:00 ryanzl1291
The rate constant for a first order reaction
is 3.5-10-3 S-1 at 25°C. The rate constant
at 77°C is 0.70 S-1. What is the activation
energy for the reaction?
[ ? ]kJ/mol
Recall that R = 8.314 J/mol K
-hurry it’s a test

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 02:00
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1

Chemistry, 22.06.2019 13:50
Amap that uses a range of colors and shading to represent the elevation, depth, or landscape of specific features on earth is a/an map.
Answers: 3

Chemistry, 23.06.2019 12:10
What is the correct name for hg(no3)2? mercury (i) nitrate mercury (ii) nitrate mercury nitroxide mercury dinitride
Answers: 1
You know the right answer?
Questions

History, 14.06.2021 22:50



History, 14.06.2021 22:50



Mathematics, 14.06.2021 22:50

Computers and Technology, 14.06.2021 22:50



Mathematics, 14.06.2021 22:50

Mathematics, 14.06.2021 22:50

Mathematics, 14.06.2021 22:50




Chemistry, 14.06.2021 22:50





