Chemistry, 01.03.2021 23:10 harveyangel123p2tjae
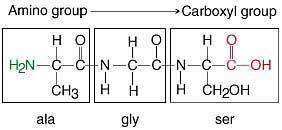
Examine the peptide. Peptide with three amino acid residues. The first residue contains a methyl group side chain. The second residue contains a hydrogen atom side chain. The third residue contains a hydroxymethyl group side chain. The amino group of the first side chain binds to the unshown remainder of the peptide. The carboxyl group of the third side chain binds to the unshown remainder of the peptide. Determine which amino acids are present in the peptide.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
Examine the peptide. Peptide with three amino acid residues. The first residue contains a methyl gro...
Questions

Biology, 30.08.2021 09:30


Advanced Placement (AP), 30.08.2021 09:30


Mathematics, 30.08.2021 09:30

Mathematics, 30.08.2021 09:30


Engineering, 30.08.2021 09:30


Mathematics, 30.08.2021 09:30


Mathematics, 30.08.2021 09:40

Computers and Technology, 30.08.2021 09:40

History, 30.08.2021 09:40

Biology, 30.08.2021 09:40



Mathematics, 30.08.2021 09:40

Health, 30.08.2021 09:40