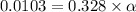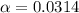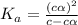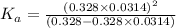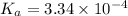Chemistry, 01.03.2021 22:20 william1690
In the laboratory, a general chemistry student measured the pH of a 0.328 M aqueous solution of acetylsalicylic acid (aspirin), HC9H7O4 to be 1.987. Use the information she obtained to determine the Ka for this acid.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:10
When the volume and number of particles of a gas are constant which of the following is also constant
Answers: 3

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

You know the right answer?
In the laboratory, a general chemistry student measured the pH of a 0.328 M aqueous solution of acet...
Questions



Mathematics, 18.10.2019 08:30


Social Studies, 18.10.2019 08:30

Chemistry, 18.10.2019 08:30

Chemistry, 18.10.2019 08:30


Mathematics, 18.10.2019 08:30




Mathematics, 18.10.2019 08:30

Mathematics, 18.10.2019 08:30

Mathematics, 18.10.2019 08:30




History, 18.10.2019 08:30

Mathematics, 18.10.2019 08:30

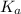 for the acid is
for the acid is 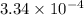
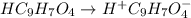
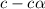
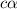
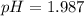
![1.987=-log[H^+]](/tpl/images/1158/1923/8f2fb.png)
![[H^+]=0.0103](/tpl/images/1158/1923/4a7f1.png)
![[H^+]=c\times \alpha](/tpl/images/1158/1923/4fc41.png)