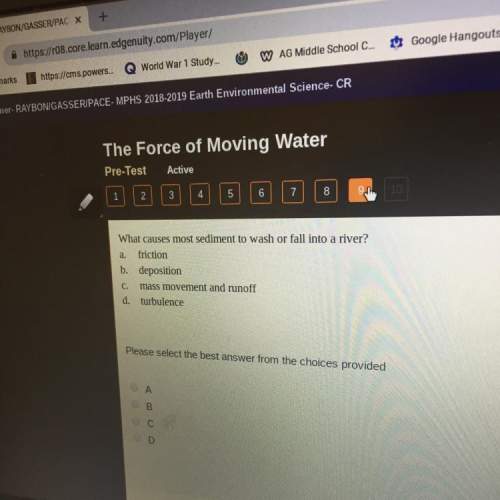
A solution is prepared by dissolving 29.6 g urea, , in 130.0 g water. Calculate the boiling point of the solution. Urea is a nonelectrolyte. (This problem requires values in your textbook's specific appendices, which you can access through the OWLv2 MindTap Reader. You should not use the OWLv2 References' Tables to answer this question as the values will not match.)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon? a balloon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1

Chemistry, 22.06.2019 15:20
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
A solution is prepared by dissolving 29.6 g urea, , in 130.0 g water. Calculate the boiling point of...
Questions

Biology, 25.08.2019 12:30




Mathematics, 25.08.2019 12:30




Mathematics, 25.08.2019 12:30

Social Studies, 25.08.2019 12:30

History, 25.08.2019 12:30

Chemistry, 25.08.2019 12:30

Health, 25.08.2019 12:30


Biology, 25.08.2019 12:30

Biology, 25.08.2019 12:30

Mathematics, 25.08.2019 12:30


Mathematics, 25.08.2019 12:30