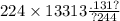Chemistry, 28.02.2021 18:50 ninaaforever
In the following take CV = 20.8 and CP = 29.1 J⋅mol−1⋅°C−1 for nitrogen gas: (a) Three moles of nitrogen at 30°C, contained in a rigid vessel, is heated to 250°C. How much heat is required if the vessel has a negligible heat capacity? If the vessel weighs 100 kg and has a heat capacity of 0.5 kJ⋅kg−1⋅°C−1, how much heat is required? (b) Four moles of nitrogen at 200°C is contained in a piston/cylinder arrangement. How much heat must be extracted from this system, which is kept at constant pressure, to cool it to 40°C if the heat capacity of the piston and cylinder is neglected?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3


Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1
You know the right answer?
In the following take CV = 20.8 and CP = 29.1 J⋅mol−1⋅°C−1 for nitrogen gas: (a) Three moles of nitr...
Questions


Biology, 19.09.2019 15:30


Mathematics, 19.09.2019 15:30


Mathematics, 19.09.2019 15:30




Social Studies, 19.09.2019 15:30

Mathematics, 19.09.2019 15:30



Mathematics, 19.09.2019 15:30

Social Studies, 19.09.2019 15:30

Mathematics, 19.09.2019 15:30

Mathematics, 19.09.2019 15:30


Biology, 19.09.2019 15:30