Chemistry, 28.02.2021 17:20 lailabirdiemae
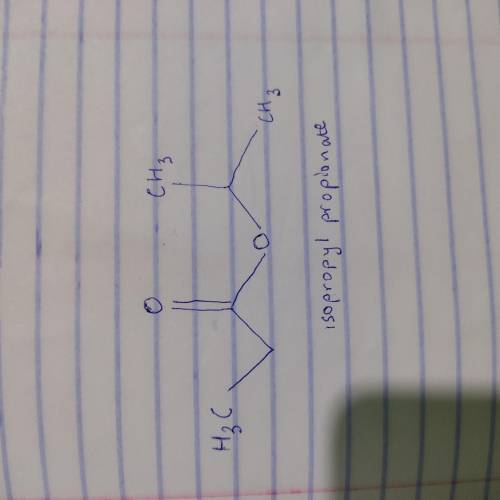
An unknown compound X has the empirical formula C3H6O and a molecular ion peak in its mass spectrum at m/z 116. X shows no IR absorption at 3200-3600 cm^-1 but shows a peak at 1740 cm^-1. The H NMR spectral data of X is shown below. What is the most likely structure of X? 22. An unknown compound X has the molecular formula C6H14O. X shows a strong peak in its IR spectrum at 3000 cm^-1. The 1H NMR spectral data of N are given below. What is the most likely structure of X?
Absorption ζ H ratio
triplet 1.0 3
double 1.0 6
quartet 2.0 2
septet 3.5 1

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2


Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
An unknown compound X has the empirical formula C3H6O and a molecular ion peak in its mass spectrum...
Questions

Computers and Technology, 13.08.2020 14:01

Mathematics, 13.08.2020 14:01

Social Studies, 13.08.2020 14:01

Chemistry, 13.08.2020 14:01

Mathematics, 13.08.2020 14:01

Chemistry, 13.08.2020 14:01




Mathematics, 13.08.2020 14:01

Geography, 13.08.2020 14:01


Mathematics, 13.08.2020 14:01




Mathematics, 13.08.2020 14:01

Mathematics, 13.08.2020 14:01

English, 13.08.2020 14:01