O A. -195 kJ

Chemistry, 26.02.2021 23:10 Mathcat444
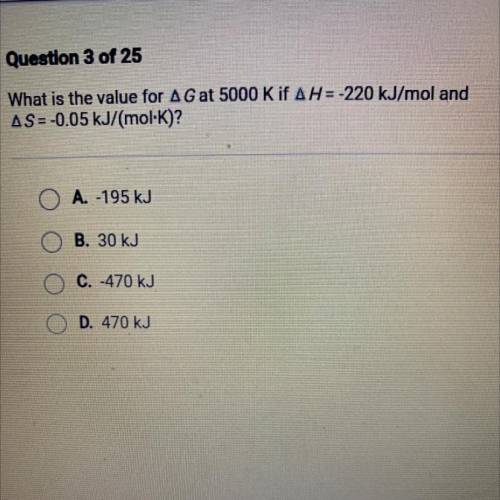
What is the value for AG at 5000 K IF AH=-220 kJ/moland
AS=-0.05 kJ/(mol·K)?
O A. -195 kJ
B. 30 kJ
C. -470 kJ
D. 470 kJ

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 01:30
Which statement accurately represents the arrangement of electrons in bohr’s atomic model?
Answers: 2

Chemistry, 23.06.2019 09:30
How many moles of na2s2o3 are needed to react with 0.12mol of cl2? show work.
Answers: 1

Chemistry, 23.06.2019 10:30
The element chlorine has two stable isotopes, chlorine-35 with a mass of 34.97 amu and chlorine-37 with a mass of 36.95 amu. from the atomic weight of cl = 35.45 one can conclude that:
Answers: 2
You know the right answer?
What is the value for AG at 5000 K IF AH=-220 kJ/moland
AS=-0.05 kJ/(mol·K)?
O A. -195 kJ
O A. -195 kJ
Questions


History, 12.11.2020 19:10

Mathematics, 12.11.2020 19:10

Biology, 12.11.2020 19:10


History, 12.11.2020 19:10


English, 12.11.2020 19:10


History, 12.11.2020 19:10



Mathematics, 12.11.2020 19:10


Mathematics, 12.11.2020 19:10




Health, 12.11.2020 19:10



