
Chemistry, 26.02.2021 04:40 gabrielleteti
How many moles of H3(PO4) are produced when 71.0 g P4O10 reacts
completely with H2O in the reaction?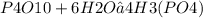

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2
You know the right answer?
How many moles of H3(PO4) are produced when 71.0 g P4O10 reacts
completely with H2O in the reaction...
Questions


Biology, 06.04.2020 23:27





Biology, 06.04.2020 23:27



Mathematics, 06.04.2020 23:27

English, 06.04.2020 23:27

Mathematics, 06.04.2020 23:27



Business, 06.04.2020 23:28


Mathematics, 06.04.2020 23:28





