
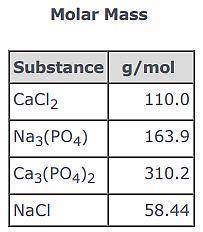
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s) + 6NaCl(aq)
Use the balanced equation and the Molar Mass table above to answer the following question.
Suppose 163.9 g of Na3(PO4) reacted with sufficient CaCl2 in solution to actually yield 116 g of Ca3(PO4)2(s) . What is the percent yield of Ca3(PO4)2(s)?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Asample of neon occupies a volume of 375 ml at stp. what will be the volume of neon if the pressure is reduced to 90.0 kpa? a. 422 ml b. 422 l c. 333 ml d. 333 l
Answers: 2


Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
3CaCl2(aq) + 2Na3(PO4)(aq) → Ca3(PO4)2(s) + 6NaCl(aq)
Use the balanced equation and the Molar Mass...
Questions

English, 02.06.2021 07:10

Mathematics, 02.06.2021 07:10

Physics, 02.06.2021 07:10

Chemistry, 02.06.2021 07:10


Biology, 02.06.2021 07:10

Chemistry, 02.06.2021 07:10

History, 02.06.2021 07:10

Computers and Technology, 02.06.2021 07:10


Mathematics, 02.06.2021 07:10

Mathematics, 02.06.2021 07:10


Mathematics, 02.06.2021 07:10

Mathematics, 02.06.2021 07:10

Mathematics, 02.06.2021 07:10


Biology, 02.06.2021 07:20


Mathematics, 02.06.2021 07:20



