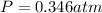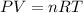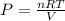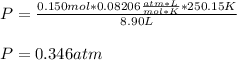Chemistry, 25.02.2021 08:30 majorsam82
Writing Sample: At what pressure would 0.150 mole of nitrogen gas at -23.0 o C occupy 8.90 L?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1

You know the right answer?
Writing Sample: At what pressure would 0.150 mole of nitrogen gas at -23.0 o C occupy 8.90 L?...
Questions














Mathematics, 31.12.2019 02:31