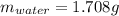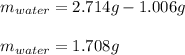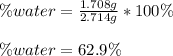A 2.714 gram sample of a hydrate of sodium carbonate is placed in a test
tube and heated until the mass is constant. If the mass of the dry sodium
carbonate is 1.006 grams. What was the mass of water driven off and what
was the percentage of water in the hydrate?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
You know the right answer?
A 2.714 gram sample of a hydrate of sodium carbonate is placed in a test
tube and heated until the...
Questions


Chemistry, 25.06.2021 14:00

Mathematics, 25.06.2021 14:00

History, 25.06.2021 14:00


Chemistry, 25.06.2021 14:00


Mathematics, 25.06.2021 14:00

History, 25.06.2021 14:00






Mathematics, 25.06.2021 14:00



Mathematics, 25.06.2021 14:00

English, 25.06.2021 14:00

Physics, 25.06.2021 14:00