
Chemistry, 24.02.2021 19:50 missyli400
Is anyone good at
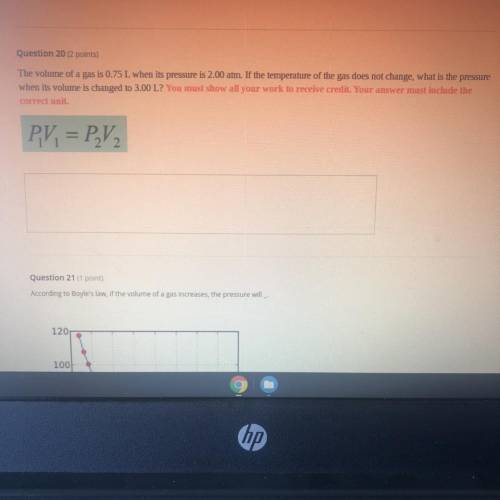
10th grade chemistry if so can you help me please ?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1
You know the right answer?
Is anyone good at
10th grade chemistry if so can you help me please ?
...
10th grade chemistry if so can you help me please ?
...
Questions


Mathematics, 26.08.2021 15:00

English, 26.08.2021 15:00


Physics, 26.08.2021 15:00




Mathematics, 26.08.2021 15:00

Mathematics, 26.08.2021 15:00

Mathematics, 26.08.2021 15:00


Mathematics, 26.08.2021 15:00



History, 26.08.2021 15:00

Social Studies, 26.08.2021 15:00

Mathematics, 26.08.2021 15:00

Physics, 26.08.2021 15:00



