
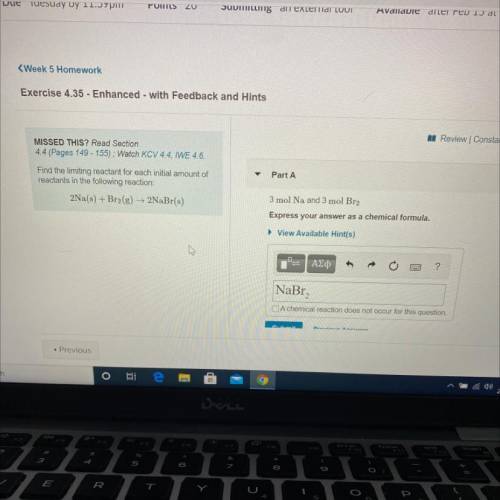
Find the limiting reactant for each initial amount of
reactants in the following reaction: 2Na(s) + Br2(g) + 2NaBr(s)
Part A: 3 mol Na and 3 mol Br2
Part B: 1.6 mol Na and 1.2 mol Br2
Part C: 2.5 mol Na and 1 mol Br2
Part D: 12.9 mol Na and 6.9 mol Br2
Express your answers as a chemical formula.
Please help! will give brainliest

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1

Chemistry, 23.06.2019 09:30
Organisms that live in the alpine and taiga biomes have developed unique adaptations that aid in their survival. moss campion is one of the plants found in the alpine biome. it has small leaves and a cushion shape that protect it from the wind and freezing temperatures in the alpine. how has the moss campion adapted to enable its survival in the alpine biome? a. waxy needles b. cone-shaped c. thin trunks d. low-growing
Answers: 1
You know the right answer?
Find the limiting reactant for each initial amount of
reactants in the following reaction: 2Na(s) +...
Questions


Mathematics, 14.08.2021 01:00



Mathematics, 14.08.2021 01:00



Mathematics, 14.08.2021 01:00





English, 14.08.2021 01:00


Mathematics, 14.08.2021 01:00


Chemistry, 14.08.2021 01:00





