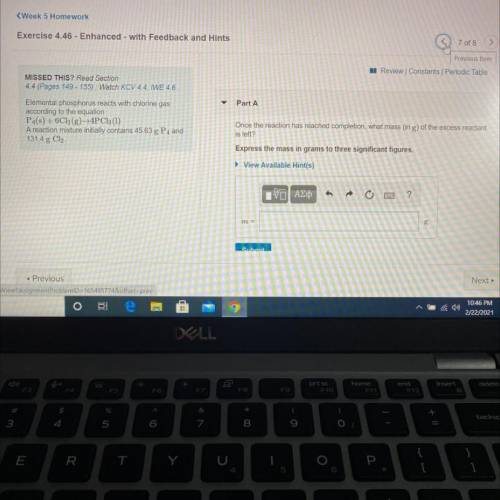
Elemental phosphorus reacts with chlorine gas
according to the equation
P4(s) + 6C12(g)+4PC13...

Chemistry, 23.02.2021 09:00 araminaara691
Elemental phosphorus reacts with chlorine gas
according to the equation
P4(s) + 6C12(g)+4PC13 (1)
A reaction mixture initially contains 45.63 g P4 and
131.4 g Cl.
Once the reaction has reached completion, what mass (in g) of the excess reactant is left?
Express the mass in grams to three significant figures.
please help i’ll give brainliest

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2


Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2
You know the right answer?
Questions










Mathematics, 31.03.2020 01:49







Mathematics, 31.03.2020 01:49


English, 31.03.2020 01:49

Mathematics, 31.03.2020 01:49



