
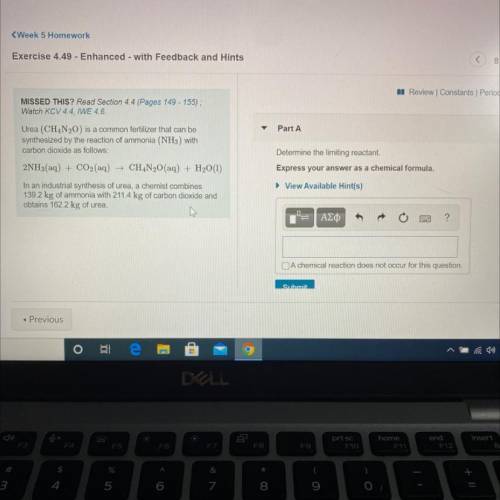
Urea (CH4N20) is a common fertilizer that can be
synthesized by the reaction of ammonia (NH3) with
carbon dioxide as follows:
Determine the limiting reactant.
Express your answer as a chemical formula.
2NH3(aq) + CO2(aq) + CH4N20(aq) + H2O(1)
In an industrial synthesis of urea, a chemist combines
139.2 kg of ammonia with 211.4 kg of carbon dioxide and
obtains 162.2 kg of urea.
Determine the limiting reactant

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2

Chemistry, 23.06.2019 07:30
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes or no?
Answers: 1

Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1
You know the right answer?
Urea (CH4N20) is a common fertilizer that can be
synthesized by the reaction of ammonia (NH3) with...
Questions

Geography, 09.10.2019 10:10






Social Studies, 09.10.2019 10:10


Social Studies, 09.10.2019 10:10

Mathematics, 09.10.2019 10:10




Mathematics, 09.10.2019 10:10





Mathematics, 09.10.2019 10:10



