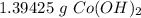Chemistry, 22.02.2021 23:30 Uhmjujiooo4220
Stoichiometry:
You conduct the following precipitation reaction in a lab:
CoCl₂ + 2NaOH → 2NaCl + Co(OH)₂
If you react 10.0 mL of 1.5 M CoCl₂ with plenty of NaOH, how many grams of Co(OH)₂ will precipitate out?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 23.06.2019 00:30
An ice cube with a volume of 45.0ml and a density of 0.9000g/cm3 floats in a liquid with a density of 1.36g/ml. what volume of the cube is submerged in the liquid
Answers: 3

Chemistry, 23.06.2019 03:00
A0.100-kilogram apple hangs in a tree 1.50 meter above the ground. ignore frictional effects, the total mechanical energy of the apples is
Answers: 1

Chemistry, 23.06.2019 04:30
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1
You know the right answer?
Stoichiometry:
You conduct the following precipitation reaction in a lab:
CoCl₂ + 2NaOH → 2Na...
CoCl₂ + 2NaOH → 2Na...
Questions

Spanish, 13.01.2021 17:50










Social Studies, 13.01.2021 17:50

Mathematics, 13.01.2021 17:50

English, 13.01.2021 17:50


Mathematics, 13.01.2021 17:50




Health, 13.01.2021 17:50

Social Studies, 13.01.2021 17:50

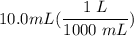 [DA] Multiply/Divide [Cancel out units]:
[DA] Multiply/Divide [Cancel out units]: 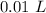 [DA] Find moles of CoCl₂ [Molarity]:
[DA] Find moles of CoCl₂ [Molarity]: 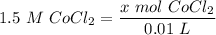 [DA] Solve for x [Multiplication Property of Equality]:
[DA] Solve for x [Multiplication Property of Equality]: 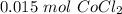 [DA] Set up [Reaction Stoich]:
[DA] Set up [Reaction Stoich]: 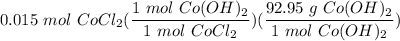 [DA] Multiply/Divide [Cancel out units]:
[DA] Multiply/Divide [Cancel out units]: