Chemistry, 22.02.2021 19:50 villarrealc1987
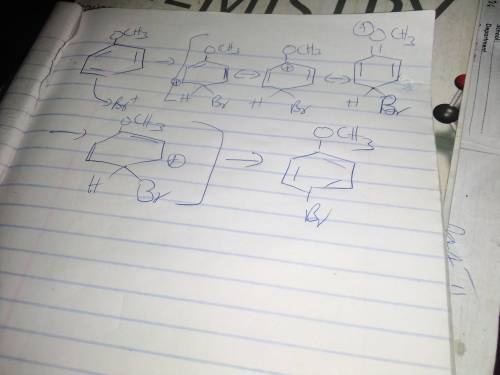
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Groups, already present on the benzene ring, that direct ortho/para further stabilize this intermediate by participating in the resonance delocalization of the positive charge. Assume that the following group is present on a benzene ring at position 1 and that you are brominating the ring at positon 4. In the box below draw the structure of the resonance contributor that shows this group actively participating in the charge delocalization. -OCH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1
You know the right answer?
During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed. Gr...
Questions



Computers and Technology, 17.01.2021 04:30


Business, 17.01.2021 04:30

Biology, 17.01.2021 04:30

Biology, 17.01.2021 04:30

Social Studies, 17.01.2021 04:30




Social Studies, 17.01.2021 04:30

Mathematics, 17.01.2021 04:30


History, 17.01.2021 04:30


Mathematics, 17.01.2021 04:30


Mathematics, 17.01.2021 04:30

English, 17.01.2021 04:30