
Chemistry, 22.02.2021 19:30 tejalawatson91
Which of the following are correct for zero-order reactions?
A. A higher concentration of reactants will not increase the reaction rate.
B. The units for the rate constant and the rate of reaction are the same.
C. The rate of reaction does not equal the rate constant.
D. The concentration of the reactants changes nonlinearly.
E. A zero-order reaction slows down as the reaction proceeds.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Asample of radium-226 will decay 1/4 of its original amount after 3200years. what is the half-life of radium-226?
Answers: 2

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3
You know the right answer?
Which of the following are correct for zero-order reactions?
A. A higher concentration of reactants...
Questions


Mathematics, 09.12.2020 23:30



Mathematics, 09.12.2020 23:30






Mathematics, 09.12.2020 23:30

Mathematics, 09.12.2020 23:30


Arts, 09.12.2020 23:30


Mathematics, 09.12.2020 23:30

Mathematics, 09.12.2020 23:30

Mathematics, 09.12.2020 23:30

Mathematics, 09.12.2020 23:30

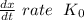 doesn't depend on reaction rate, a higher reaction rate does not intensify the reaction.
doesn't depend on reaction rate, a higher reaction rate does not intensify the reaction.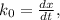 the created based and the reaction rate is about the same.
the created based and the reaction rate is about the same.

